Abstract
Overexpression of the HMGA2 gene is a common feature of neoplastic cells both in experimental and human models. Intragenic and extragenic HMGA2 rearrangements responsible for HMGA2 gene overexpression have been frequently detected in human benign tumours of mesenchymal origin. To better understand the role of HMGA2 overexpression in human tumorigenesis, we have generated transgenic mice carrying the HMGA2 gene under the transcriptional control of the cytomegalovirus promoter. High expression of the transgene was demonstrated in all the mouse tissues analysed, whereas no expression of the endogenous HMGA2 gene was detected in the same tissues from wild-type mice. In this study, two indipendent lines of transgenic mice have been generated. By 6 months of age, 85% of female animals of both transgenic lines developed pituitary adenomas secreting prolactin and growth hormone. The transgenic males developed the same phenotype with a lower penetrance (40%) and a longer latency period (about 18 months). Therefore, these data demonstrate that the overexpression of HMGA2 leads to the onset of mixed growth hormone/prolactin cell pituitary adenomas. These transgenic mice may represent an important tool for the study of this kind of neoplasia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
09 December 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41388-022-02535-z
References
Abe N, Watanabe T, Sugiyama M, Uchimura H, Chiappetta G, Fusco A, Atomi Y . 1999 Cancer Res. 59: 1169–1174
Abe N, Watanabe T, Masaki T, Mori T, Sugiyama M, Uchimura H, Fujioka Y, Chiappetta G, Fusco A, Atomi Y . 2000 Cancer Res. 60: 3117–3122
Anand A, Chada K . 2000 Nat. Genet 24: 377–380
Arlotta P, Tai AK, Manfioletti G, Clifford C, Jay G, Ono SJ . 2000 J. Biol. Chem. 275: 14394–14400
Ashar HR, Fejzo MS, Tkachenko A, Zhou X, Fletcher JA, Weremowicz S, Morton CC, Chada K . 1995 Cell 82: 57–65
Bandiera A, Bonifacio D, Manfioletti G, Mantovani F, Rustighi A, Zanconati F, Fusco A, Di Bonito L, Giancotti V . 1998 Cancer Res. 58: 426–431
Battista S, Fidanza V, Fedele M, Klein-Szanto AJP, Outwater E, Brunner H, Santoro M, Croce CM, Fusco A . 1999 Cancer Res. 59: 4793–4797
Bauer K, Carmeliet P, Schulz M, Baes M, Deuef C . 1990 Endocrinology 127: 1224–1233
Berlingieri MT, Manfioletti G, Santoro M, Bandiera A, Visconti R, Giancotti V, Fusco A . 1995 Mol. Cell. Biol. 15: 1545–1553
Bol S, Wanschura S, Thode B, Deichert U, Van de Ven WJ, Bartnitzke S, Bullerdiek J . 1996 Cancer Genet. Cytogenet. 90: 88–90
Chiappetta G, Bandiera A, Berlingieri MT, Visconti R, Manfioletti G, Battista S, Martinez-Tello FJ, Santoro M, Giancotti V, Fusco A . 1995 Oncogene 10: 1307–1314
Chiappetta G, Avantaggiato V, Visconti R, Fedele M, Battista S, Trapasso F, Merciai BM, Fidanza V, Giancotti V, Santoro M, Simeone A, Fusco A . 1996 Oncogene 13: 2439–2446
Chiappetta G, Tallini G, De Biasio MC, Manfioletti G, Martinez-Tello FJ, Pentimalli F, de Nigris F, Mastro A, Botti G, Fedele M, Berger N, Santoro M, Giancotti V, Fusco A . 1998 Cancer Res. 58: 4193–4198
Chiappetta G, Manfioletti G, Pentimalli F, Abe N, Di Bonito M, Vento MT, Giuliano A, Fedele M, Viglietto G, Santoro M, Watanabe T, Giancotti V, Fusco A . 2001 Int. J. Cancer 91: 147–151
Fedele M, Bandiera A, Chiappetta G, Battista S, Viglietto G, Manfioletti G, Casamassimi A, Santoro M, Giancotti V, Fusco A . 1996 Cancer Res. 56: 1896–1901
Fedele M, Berlingieri MT, Scala S, Chiariotti L, Viglietto G, Rippel V, Bullerdiek J, Santoro M, Fusco A . 1998 Oncogene 17: 413–418
Fedele M, Battista S, Manfioletti G, Croce CM, Giancotti V, Fusco A . 2001 Carcinogenesis 22: 1583–1591
Finelli P, Giardino D, Rizzi N, Buiatiotis S, Virduci T, Franzin A, Losa M, Larizza L . 2000 Int. J. Cancer 86: 344–350
Finelli P, Pierantoni GM, Giardino D, Losa M, Rodeschini O, Fedele M, Valtorta E, Mortini P, Croce CM, Larizza L, Fusco A . 2002 Cancer Res. in press
Giancotti V, Pani B, D'Andrea P, Berlingieri MT, Di Fiore PP, Fusco A, Vecchio G, Philp R, Crane-Robinson C, Nicolas RH, Wright CA, Goodwin GH . 1987 EMBO J. 6: 1981–1987
Hogan B . 1983 Nature 306: 313–314
Johnson KR, Lehn DA, Reeves R . 1989 Mol. Cell. Biol. 9: 2114–2123
Kazmierczak B, Wanschura S, Rosigkeit J, Mejer-Bolte K, Uschinsky K, Hanpt R, Schoenmakers EFPM, Barnitzke S, Van de Ven W, Bullerdiek J . 1995 Cancer Res. 55: 2497–2499
Nagpal S, Ghosn C, DiSepio D, Molina Y, Sutter M, Klein ES, Chandraratna RAS . 1999 J. Biol. Chem. 274: 22563–22568
Pellegrini-Bouiller I, Morange-Ramos I, Barlier A, Gunz G, Enjalbert A, Jaquet P . 1997 Horm. Res. 47: 251–258
Rogalla P, Drechsler K, Frey G, Hennig Y, Helmke B, Bonk U, Bullerdiek J . 1996 Am. J. Pathol. 149: 775–779
Rommel B, Rogalla P, Jox A, Kalle CV, Kazmierczak B, Wolf J, Bullerdiek J . 1997 26: 603–607
Schoenberg Fejzo M, Ashar HR, Krauter KS, Powell WL, Rein MS, Weremowicz S, Yoon SJ, Kucherlapati RS, Chada K, Morton CC . 1996 Genes Chrom. Cancer 17: 1–6
Schoenmakers EFPM, Wanschura S, Mols R, Bullerdiek J, Van den Berghe H, Van de Ven WJ . 1995 Nat. Genet. 10: 436–444
Spady TJ, McComb RD, Shull JD . 1999 Endocrine 11: 217–233
Staats B, Wanschura S, Hanisch P, Schoenmakers EF, Van de Ven WJ, Bartnitzke S, Bullerdiek J . 1996 Breast Cancer Res. Treat. 38: 299–303
Thanos D, Maniatis T . 1995 Cell 83: 1091–1100
Tsokos M, Scarpa S, Ross RA, Triche TJ . 1987 Am. J. Pathol. 128: 484–496
Weil RJ, Huang S, Pack S, Vortmeyer AO, Tsokos M, Lubensky IA, Oldfield EH, Zhuang Z . 1998 Cancer Res. 58: 4715–4720
Wood LJ, Maher JF, Bunton TE, Resar LM . 2000 Cancer Res. 60: 4256–4261
Zhou X, Benson KF, Ashar HR, Chada K . 1995 Nature 376: 771–774
Acknowledgements
We thank Andrea Vecchione for useful discussions about the autoptic analysis of mice, Chris Munnery for the technical expertise in performing the in situ double immunofluorescence staining and the Associazione Partenopea per le Ricerche Oncologiche (APRO) for its support. This work was supported by grants P01CA76259 and P30CA56036 from the National Cancer Institute to CM Croce and by grants from AIRC and MURST, projects ‘Terapie antineoplastiche innovative’ and ‘Piani di Potenziamento della Rete Scientifica e Tecnologica’. GM Pierantoni was supported by a fellowship from the FIRC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fedele, M., Battista, S., Kenyon, L. et al. Overexpression of the HMGA2 gene in transgenic mice leads to the onset of pituitary adenomas. Oncogene 21, 3190–3198 (2002). https://doi.org/10.1038/sj.onc.1205428
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205428
Keywords
This article is cited by
-
Genomics and Epigenomics of Pituitary Tumors: What Do Pathologists Need to Know?
Endocrine Pathology (2021)
-
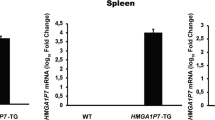
HMGA1-pseudogene7 transgenic mice develop B cell lymphomas
Scientific Reports (2020)
-
RPSAP52 lncRNA is overexpressed in pituitary tumors and promotes cell proliferation by acting as miRNA sponge for HMGA proteins
Journal of Molecular Medicine (2019)
-
mTOR promotes pituitary tumor development through activation of PTTG1
Oncogene (2017)
-
Oncofetal HMGA2 effectively curbs unconstrained (+) and (−) DNA supercoiling
Scientific Reports (2017)