Abstract
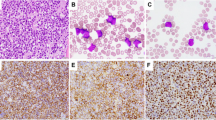
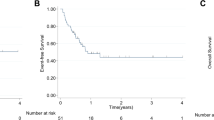
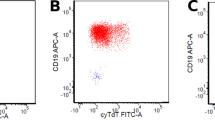
To evaluate the frequency and cytogenetic and immunophenotypic features of therapy-related, precursor B-cell acute lymphoblastic leukemia (ALL), 152 cases of immature B-cell ALL were reviewed. These were compared to the frequency of therapy-related acute myeloid leukemia (t-AML) during the same time period. Eight ALL cases with a prior diagnosis of malignancy were identified, including six (4.0%) with prior therapy considered to be therapy-related ALL (t-ALL). The t-ALL cases followed treatment for breast carcinoma (two cases), lung carcinoma (two cases), lymphocyte predominance Hodgkin's disease and follicular lymphoma with a latency period of 13 months to 8 years. All t-ALL cases had a pro-B (CD10-negative) immunophenotype with significantly higher expression of CD15 and CD65, compared to the de novo CD10-positive ALL cases. All six t-ALL cases had MLL abnormalities by fluorescence in situ hybridization, and four showed t(4;11)(q21;q23). These represented half of all 11q23-positive adult ALL cases. During the same time period, 4.9% of all AML cases were considered t-AML. There was a 16.7% frequency of 11q23 abnormalities in the t-AML group. Despite the similar frequency in therapy-related disease among ALL and AML cases, there were differences in the frequency of the diseases and t-ALL represented 12% of all therapy-related leukemias. However, t-ALL represented 46% of all 11q23-positive therapy-related leukemias. The immunogenetic features of t-ALL appear distinct and may aid in identifying more cases of this disease type in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Pui C-H, Behm FG, Raimondi SC, Dodge RK, George SL, Rivera GK et al. Secondary acute myeloid leukemia in children treated for acute lymphoid leukemia. N Engl J Med 1989; 321: 136–142.
Bhatia S, Ramsay NKC, Steinbuch M, Dusenbery KE, Shapiro RS, Weisdorf DJ et al. Malignant neoplasms following bone marrow transplantation. Blood 1996; 87: 3633–3639.
Pagano L, Pulsoni A, Tosti ME, Avvisati G, Mele L, Mele A et al. Clinical and biological features of acute myeloid leukaemia occurring as second malignancy: GIMEMA archive of adult acute leukaemia. Br J Haematol 2001; 112: 109–117.
Bhatia S, Louie AD, Bhatia R, O'Donnell MR, Fung H, Kashyap A et al. Solid cancers after bone marrow transplantation. J Clin Oncol 2001; 19: 464–471.
Rowley JD, Olney HJ . International workshop on the relationship of prior therapy to balanced chromosome aberrations in therapy-related myelodysplastic syndromes and acute leukemia: overview report. Genes Chromosomes Cancer 2002; 33: 331–345.
Traweek ST, Slovak ML, Nademanee AP, Brynes RK, Niland JC, Forman SJ . Clonal karyotypic hematopoietic-cell abnormalities occurring after autologous bone-marrow transplantation for Hodgkin's-disease and non-Hodgkin's-lymphoma. Blood 1994; 84: 957–963.
Pagano L, Pulsoni A, Tosti ME, Annino L, Mele A, Camera A et al. Acute lymphoblastic leukaemia occurring as second malignancy: report of the GIMEMA archive of adult acute leukaemia. Gruppo Italiano Malattie Ematologiche Maligne dell’Adulto. Br J Haematol 1999; 106: 1037–1040.
Andersen MK, Christiansen DH, Jensen BA, Ernst P, Hauge G, Pedersen-Bjergaard J . Therapy-related acute lymphoblastic leukaemia with MLL rearrangements following DNA topoisomerase II inhibitors, an increasing problem: report on two new cases and review of the literature since 1992. Br J Haematol 2001; 114: 539–543.
Bloomfield CD, Archer KJ, Mrozek K, Lillington DM, Kaneko Y, Head DR et al. 11q23 balanced chromosome aberrations in treatment-related myelodysplastic syndromes and acute leukemia: report from an international workshop. Genes Chromosomes Cancer 2002; 33: 362–378.
Khalidi HS, Chang KL, Medeiros LJ, Brynes RK, Slovak ML, Murata-Collins J et al. Acute lymphoblastic leukemia. Survey of immunophenotype, French–American–British classification, frequency of myeloid antigen expression, and karyotypic abnormalities in 210 pediatric and adult cases. Am J Clin Pathol 1999; 111: 467–476.
Arber DA, Jenkins KA . Paraffin section immunophenotyping of acute leukemias in bone marrow specimens. Am J Clin Pathol 1996; 106: 462–468.
ISCN (1995): In: Mitelman F (ed.), An International System for Human Cytogenetic Nomenclature. Basel: Karger, 1995.
Snyder DS, Rossi JJ, Wang J-J, Sniecinski IJ, Slovak ML, Wallace RB et al. Persistence of bcr-abl gene expession following bone marrow transplantation for chronic myelogenous leukemia in chronic phase. Transplantation 1991; 51: 1033–1040.
Pedersen-Bjergaard J . Acute lymphoid leukemia with t(4;11) (q21;q23) following chemotherapy with cytostatic agents targeting at DNA-topoisomerase II. Leukemia Res 1992; 16: 733–735.
Block AW, Carroll AJ, Hagemeijer A, Michaux L, van Lom K, Olney HJ et al. Rare recurring balanced chromosome abnormalities in therapy-related myelodysplastic syndromes and acute leukemia: report from an international workshop. Genes Chromosomes Cancer 2002; 33: 401–412.
Secker-Walker LM, Moorman AV, Bain BJ, Mehta AB . Secondary acute leukemia and myelodysplastic syndrome with 11q23 abnormalities. Leukemia 1998; 12: 840–844.
Pui CH, Rubnitz JE, Hancock ML, Downing JR, Raimondi SC, Rivera GK et al.. Reappraisal of the clinical and biologic significance of myeloid- associated antigen expression in childhood acute lymphoblastic leukemia. J Clin Oncol 1998; 16: 3768–3773.
Lenormand B, Bene MC, Lesesve JF, Bastard C, Tilly H, Lefranc MP et al. PreB1 (CD10−) acute lymphoblastic leukemia: immunophenotypic and genomic characteristics, clinical features and outcome in 38 adults and 26 children. The Groupe dEtude Immunologique des Leucemies. Leukemia Lymphoma 1998; 28: 329–342.
Ludwig WD, Rieder H, Bartram CR, Heinze B, Schwartz S, Gassmann W et al. Immunophenotypic and genotypic features, clinical characteristics, and treatment outcome of adult pro-B acute lymphoblastic leukemia: results of the German multicenter trials GMALL 03/87 and 04/89. Blood 1998; 92: 1898–1909.
Borkhardt A, Wuchter C, Viehmann S, Pils S, Teigler-Schlegel A, Stanulla M et al. Infant acute lymphoblastic leukemia–combined cytogenetic, immunophenotypical and molecular analysis of 77 cases. Leukemia 2002; 16: 1685–1690.
Cimino G, Rapanotti MC, Biondi A, Elia L, LoCoco F, Price C et al. Infant acute leukemias show the same biased distribution of ALL1 gene breaks as topoisomerase II related secondary acute leukemias. Cancer Res 1997; 57: 2879–2883.
Acknowledgements
This work was supported in part by NIH CA-33572 and CA-30206.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ishizawa, S., Slovak, M., Popplewell, L. et al. High frequency of pro-B acute lymphoblastic leukemia in adults with secondary leukemia with 11q23 abnormalities. Leukemia 17, 1091–1095 (2003). https://doi.org/10.1038/sj.leu.2402918
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402918