Key Points
-
TWEAK (tumour necrosis factor (TNF)-like weak inducer of apoptosis) is a member of the TNF superfamily of structurally related cytokines. It is present on the cell surface but a soluble form can also be produced by proteolytic cleavage within the stalk domain. TWEAK gene expression has been detected in many cell types and tissues.
-
TWEAK binds to Fn14 (fibroblast growth factor-inducible 14), a small (102 amino acids) plasma-membrane-anchored receptor. The Fn14 gene is a growth-factor-inducible, immediate-early response gene, and it is highly expressed following tissue injury and in numerous solid tumour types.
-
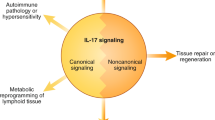
TWEAK treatment of cells in culture activates several intracellular signalling pathways, including the nuclear factor-κB (NF-κB) pathway, and induces various cellular responses. TWEAK is a pro-angiogenic and pro-inflammatory cytokine in vivo.
-
Persistent TWEAK–Fn14 engagement or Fn14 overexpression may contribute to acute ischaemic stroke, rheumatoid arthritis, systemic lupus erythematosus, multiple sclerosis and cancer.
-
The more unique and also puzzling aspects of TWEAK and Fn14 biology are discussed. In particular, emphasis will be on recent findings supporting the notion that this ligand–receptor axis might contribute to tissue repair after injury and the pathogenesis of several diseases that are responsible for significant morbidity and mortality worldwide. Strategies that may be useful for therapeutically targeting this axis will also be discussed.
Abstract
TWEAK is a multifunctional cytokine that controls many cellular activities including proliferation, migration, differentiation, apoptosis, angiogenesis and inflammation. TWEAK acts by binding to Fn14, a highly inducible cell-surface receptor that is linked to several intracellular signalling pathways, including the nuclear factor-κB (NF-κB) pathway. The TWEAK–Fn14 axis normally regulates various physiological processes, in particular it seems to play an important, beneficial role in tissue repair following acute injury. Furthermore, recent studies have indicated that TWEAK–Fn14 axis signalling may contribute to cancer, chronic autoimmune diseases and acute ischaemic stroke. This Review provides an overview of TWEAK–Fn14 axis biology and summarizes the available data supporting the proposal that both TWEAK and Fn14 should be considered as potential targets for the development of novel therapeutics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Foster, D., Parrish-Novak, J., Fox, B. & Xu, W. Cytokine–receptor pairing: accelerating discovery of cytokine function. Nature Rev. Drug Discov. 3, 160–170 (2004).
Locksley, R. M., Killeen, N. & Lenardo, M. J. The TNF and TNF receptor superfamilies: integrating mammalian biology. Cell 104, 487–501 (2001).
Bodmer, J., Schneider, P. & Tschopp, J. The molecular architecture of the TNF superfamily. Trends Biochem. Sci. 27, 19–26 (2002).
Chicheportiche, Y. et al. TWEAK, a new secreted ligand in the tumor necrosis factor family that weakly induces apoptosis. J. Biol. Chem. 272, 32401–32410 (1997). References 4 and 5 describe the initial cloning and characterization of the TWEAK cytokine.
Marsters, S. A. et al. Identification of a ligand for the death-domain-containing receptor Apo3. Curr. Biol. 8, 525–528 (1998).
Schneider, P. et al. TWEAK can induce cell death via endogenous TNF and TNF receptor 1. Eur. J. Immunol. 29, 1785–1792 (1999).
Kaptein, A. et al. Studies on the interaction between TWEAK and the death receptor WSL-1/TRAMP (DR3). FEBS Lett. 485, 135–141 (2000).
Lynch, C. N. et al. TWEAK induces angiogenesis and proliferation of endothelial cells. J. Biol. Chem. 274, 8455–8459 (1999).
Wiley, S. R. et al. A novel TNF receptor family member binds TWEAK and is implicated in angiogenesis. Immunity 15, 837–846 (2001). A pivotal paper describing the successful cloning of a TWEAK-binding cell surface protein (TWEAKR, Fn14) from an endothelial cell cDNA expression library.
Meighan-Mantha, R. L. et al. The mitogen-inducible Fn14 gene encodes a type I transmembrane protein that modulates fibroblast adhesion and migration. J. Biol. Chem. 274, 33166–33176 (1999).
Feng, S. Y. et al. The Fn14 immediate-early response gene is induced during liver regeneration and highly expressed in both human and murine hepatocellular carcinomas. Am. J. Pathol. 156, 1253–1261 (2000). An initial report demonstrating Fn14 overexpression in human tumour specimens.
Bossen, C. et al. Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J. Biol. Chem. 281, 13964–13971 (2006).
Inman, G. J. & Allday, M. J. Apoptosis induced by TGF-β1 in Burkitt's lymphoma cells is caspase 8 dependent but is death receptor independent. J. Immunol. 165, 2500–2510 (2000).
Kaplan, M. J., Ray, D., Mo, R. R., Yung, R. L. & Richardson, B. C. TRAIL (Apo2 ligand) and TWEAK (Apo3 ligand) mediate CD4+ T cell killing of antigen-presenting macrophages. J. Immunol. 164, 2897–2904 (2000).
Borysenko, C. W. et al. Death receptor-3 mediates apoptosis in human osteoblasts under narrowly regulated conditions. J. Cell. Physiol. 209, 1021–1028 (2006).
Vince, J. E. & Silke, J. TWEAK shall inherit the earth. Cell Death Differ. 13, 1842–1844 (2006).
Burkly, L. C., Michaelson, J. S., Hahm, K., Jakubowski, A. & Zheng, T. S. TWEAKing tissue remodeling by a multifunctional cytokine: Role of TWEAK/Fn14 pathway in health and disease. Cytokine 40, 1–16 (2007). An excellent review article on the TWEAK–Fn14 pathway.
De, K. A. et al. Involvement of GSK-3β in TWEAK-mediated NF-κB activation. FEBS Lett. 566, 60–64 (2004).
Baxter, F. O. et al. IKKβ/2 induces TWEAK and apoptosis in mammary epithelial cells. Development 133, 3385–3394 (2006).
Kawakita, T. et al. Functional expression of TWEAK in human colonic adenocarcinoma cells. Int. J. Oncol. 26, 87–93 (2005).
Nakayama, M., Harada, N., Okumura, K. & Yagita, H. Characterization of murine TWEAK and its receptor (Fn14) by monoclonal antibodies. Biochem. Biophys. Res. Commun. 306, 819–825 (2003).
Chicheportiche, Y., Fossati-Jimack, L., Moll, S., Ibnou-Zekri, N. & Izui, S. Down-regulated expression of TWEAK mRNA in acute and chronic inflammatory pathologies. Biochem. Biophys. Res. Commun. 279, 162–165 (2000).
Ho, D. H. et al. Soluble tumor necrosis factor-like weak inducer of apoptosis overexpression in HEK293 cells promotes tumor growth and angiogenesis in athymic nude mice. Cancer Res. 64, 8968–8972 (2004). An initial report indicating that TWEAK could contribute to tumour angiogenesis.
De, A. et al. Intraovarian tumor necrosis factor-related weak inducer of apoptosis/fibroblast growth factor-inducible-14 ligand-receptor system limits ovarian preovulatory follicles from excessive luteinization. Mol. Endocrinol. 20, 2528–2538 (2006).
Chacon, M. R. et al. Expression of TWEAK and its receptor Fn14 in human subcutaneous adipose tissue. Relationship with other inflammatory cytokines in obesity. Cytokine 33, 129–137 (2006).
Kawakita, T. et al. Functional expression of TWEAK in human hepatocellular carcinoma: possible implication in cell proliferation and tumor angiogenesis. Biochem. Biophys. Res. Commun. 318, 726–733 (2004).
Tran, N. L. et al. The human Fn14 receptor gene is up-regulated in migrating glioma cells in vitro and overexpressed in advanced glial tumors. Am. J. Pathol. 162, 1313–1321 (2003).
Yepes, M. et al. A soluble Fn14-Fc decoy receptor reduces infarct volume in a murine model of cerebral ischemia. Am. J. Pathol. 166, 511–520 (2005).
Desplat-Jego, S. et al. TWEAK is expressed by glial cells, induces astrocyte proliferation and increases EAE severity. J. Neuroimmunol. 133, 116–123 (2002).
Maecker, H. et al. TWEAK attenuates the transition from innate to adaptive immunity. Cell 123, 931–944 (2005). The first publication that used Tweak -null mice.
Girgenrath, M. et al. TWEAK, via its receptor Fn14, is a novel regulator of mesenchymal progenitor cells and skeletal muscle regeneration. EMBO J. 25, 5826–5839 (2006).
Nakayama, M., Kayagaki, N., Yamaguchi, N., Okumura, K. & Yagita, H. Involvement of TWEAK in interferon γ-stimulated monocyte cytotoxicity. J. Exp. Med. 192, 1373–1380 (2000).
Potrovita, I. et al. Tumor necrosis factor-like weak inducer of apoptosis-induced neurodegeneration. J. Neurosci. 24, 8237–8244 (2004). An initial report linking TWEAK activity to ischaemia-induced cerebral tissue damage in a murine model of stroke.
Justo, P. et al. Cytokine cooperation in renal tubular cell injury: the role of TWEAK. Kidney Int. 70, 1750–1758 (2006).
Polek, T. C., Talpaz, M., Darnay, B. G. & Spivak-Kroizman, T. TWEAK mediates signal transduction and differentiation of RAW264.7 cells in the absence of Fn14/TweakR. Evidence for a second TWEAK receptor. J. Biol. Chem. 278, 32317–32323 (2003).
Bover, L. C. et al. A previously unrecognized protein–protein interaction between TWEAK and CD163: potential biological implications. J. Immunol. 178, 8183–8194 (2007).
Glenney, G. W. & Wiens, G. D. Early diversification of the TNF superfamily in teleosts: genomic characterization and expression analysis. J. Immunol. 178, 7955–7973 (2007).
Brown, S. A. N., Hanscom, H. N., Vu, H., Brew, S. A. & Winkles, J. A. TWEAK binding to the Fn14 cysteine-rich domain depends on charged residues located in both the A1 and D2 modules. Biochem. J. 397, 297–304 (2006).
Nakayama, M. et al. Multiple pathways of TWEAK-induced cell death. J. Immunol. 168, 734–743 (2002).
Peter, M. E. & Krammer, P. H. The CD95(APO-1/Fas) DISC and beyond. Cell Death Differ. 10, 26–35 (2003).
Wilson, C. A. & Browning, J. L. Death of HT29 adenocarcinoma cells induced by TNF family receptor activation is caspase-independent and displays features of both apoptosis and necrosis. Cell Death Differ. 9, 1321–1333 (2002).
Nakayama, M. et al. Fibroblast growth factor-inducible 14 mediates multiple pathways of TWEAK-induced cell death. J. Immunol. 170, 341–348 (2003).
Donohue, P. J. et al. TWEAK is an endothelial cell growth and chemotactic factor that also potentiates FGF-2 and VEGF-A mitogenic activity. Arterioscler. Thromb. Vasc. Biol. 23, 594–600 (2003).
Harada, N. et al. Pro-inflammatory effect of TWEAK/Fn14 interaction on human umbilical vein endothelial cells. Biochem. Biophys. Res. Commun. 299, 488–493 (2002).
Dogra, C., Changotra, H., Mohan, S. & Kumar, A. Tumor necrosis factor-like weak inducer of apoptosis inhibits skeletal myogenesis through sustained activation of nuclear factor-κB and degradation of myoD protein. J. Biol. Chem. 281, 10327–10336 (2006).
Dogra, C., Hall, S. L., Wedhas, N., Linkhart, T. A. & Kumar, A. Fibroblast growth factor inducible 14 (Fn14) is required for the expression of myogenic regulatory factors and differentiation of myoblasts into myotubes. Evidence for TWEAK-independent functions of Fn14 during myogenesis. J. Biol. Chem. 282, 15000–15010 (2007).
Perper, S. J. et al. TWEAK is a novel arthritogenic mediator. J. Immunol. 177, 2610–2620 (2006). An initial report linking TWEAK activity to tissue inflammation, angiogenesis and damage in a murine model of rheumatoid arthritis.
Kingsley, L. A., Fournier, P. G., Chirgwin, J. M. & Guise, T. A. Molecular biology of bone metastasis. Mol. Cancer Ther. 6, 2609–2617 (2007).
Jakubowski, A. et al. TWEAK induces liver progenitor cell proliferation. J. Clin. Invest. 115, 2330–2340 (2005). The first publication that used Fn14 -null mice.
Zhao, Z. et al. TWEAK/Fn14 interactions are instrumental in the pathogenesis of nephritis in the chronic graft-versus-host model of systemic lupus erythematosus. J. Immunol. 179, 7949–7958 (2007).
Polavarapu, R., Gongora, M. C., Winkles, J. A. & Yepes, M. Tumor necrosis factor-like weak inducer of apoptosis increases the permeability of the neurovascular unit through nuclear factor-κB pathway activation. J. Neurosci. 25, 10094–10100 (2005).
Zhang, X. et al. TWEAK–Fn14 pathway inhibition protects the integrity of the neurovascular unit during cerebral ischemia. J. Cereb. Blood Flow Metab. 27, 534–544 (2007).
Charge, S. B. & Rudnicki, M. A. Cellular and molecular regulation of muscle regeneration. Physiol. Rev. 84, 209–238 (2004).
Dogra, C. et al. TNF-related weak inducer of apoptosis (TWEAK) is a potent skeletal muscle-wasting cytokine. FASEB J. 21, 1857–1869 (2007).
Brown, S. A. N., Richards, C. M., Hanscom, H. N., Feng, S. L. & Winkles, J. A. The Fn14 cytoplasmic tail binds tumour-necrosis-factor-receptor-associated factors 1, 2, 3 and 5 and mediates nuclear factor-κB activation. Biochem. J. 371, 395–403 (2003).
Han, S. et al. TNF-related weak inducer of apoptosis receptor, a TNF receptor superfamily member, activates NF-κB through TNF receptor-associated factors. Biochem. Biophys. Res. Commun. 305, 789–796 (2003).
Tanabe, K., Bonilla, I., Winkles, J. A. & Strittmatter, S. M. Fibroblast growth factor-inducible-14 is induced in axotomized neurons and promotes neurite outgrowth. J. Neurosci. 23, 9675–9686 (2003).
Tran, N. L. et al. Increased fibroblast growth factor-inducible 14 expression levels promote glioma cell invasion via Rac1 and nuclear factor-κB and correlate with poor patient outcome. Cancer Res. 66, 9535–9542 (2006). An important paper linking high Fn14 levels in advanced brain tumours to poor patient outcome.
Wiley, S. R. & Winkles, J. A. TWEAK, a member of the TNF superfamily, is a multifunctional cytokine that binds the TweakR/Fn14 receptor. Cytokine Growth Factor Rev. 14, 241–249 (2003).
Fischer, D., Petkova, V., Thanos, S. & Benowitz, L. I. Switching mature retinal ganglion cells to a robust growth state in vivo: gene expression and synergy with RhoA inactivation. J. Neurosci. 24, 8726–8740 (2004).
Wang, S. et al. Transcriptional profiling suggests that Barrett's metaplasia is an early intermediate stage in esophageal adenocarcinogenesis. Oncogene 25, 3346–3356 (2006).
Dvorak, H. F. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N. Engl. J. Med. 315, 1650–1659 (1986).
Iyer, V. R. et al. The transcriptional program in the response of human fibroblasts to serum. Science 283, 83–87 (1999).
Chang, H. Y. et al. Gene expression signature of fibroblast serum response predicts human cancer progression: similarities between tumors and wounds. PLoS Biol. 2, e7 (2004).
Chang, H. Y. et al. Robustness, scalability, and integration of a wound-response gene expression signature in predicting breast cancer survival. Proc. Natl Acad. Sci. USA 102, 3738–3743 (2005).
Saitoh, T. et al. TWEAK induces NF-κB2 p100 processing and long lasting NF-κB activation. J. Biol. Chem. 278, 36005–36012 (2003).
Perkins, N. D. Integrating cell-signalling pathways with NF-κB and IKK function. Nature Rev. Mol. Cell Biol. 8, 49–62 (2007).
Hayden, M. S., West, A. P. & Ghosh, S. NF-κB and the immune response. Oncogene 25, 6758–6780 (2006).
Karin, M. Nuclear factor-kappaB in cancer development and progression. Nature 441, 431–436 (2006).
Neumann, M. & Naumann, M. Beyond IκBs: alternative regulation of NF-κB activity. FASEB J. 21, 2642–2654 (2007).
Tran, N. L. et al. The tumor necrosis factor-like weak inducer of apoptosis (TWEAK)-fibroblast growth factor-inducible 14 (Fn14) signaling system regulates glioma cell survival via NF-κB pathway activation and BCL-XL/BCL-W expression. J. Biol. Chem. 280, 3483–3492 (2005).
Campbell, S. et al. Proinflammatory effects of TWEAK/Fn14 interactions in glomerular mesangial cells. J. Immunol. 176, 1889–1898 (2006).
Ando, T. et al. TWEAK/Fn14 interaction regulates RANTES production, BMP-2-induced differentiation, and RANKL expression in mouse osteoblastic MC3T3-E1 cells. Arthritis Res. Ther. 8, R146 (2006).
Jin, L. et al. Induction of RANTES by TWEAK/Fn14 interaction in human keratinocytes. J. Invest. Dermatol. 122, 1175–1179 (2004).
Xu, H. et al. TWEAK/Fn14 interaction stimulates human bronchial epithelial cells to produce IL-8 and GM-CSF. Biochem. Biophys. Res. Commun. 318, 422–427 (2004).
Kim, S. H. et al. TWEAK can induce pro-inflammatory cytokines and matrix metalloproteinase-9 in macrophages. Circ. J. 68, 396–399 (2004).
Michaelson, J. S. et al. Tweak induces mammary epithelial branching morphogenesis. Oncogene 24, 2613–2624 (2005).
Nathan, C. Points of control in inflammation. Nature 420, 846–852 (2002).
Luster, A. D., Alon, R. & von Andrian, U. H. Immune cell migration in inflammation: present and future therapeutic targets. Nature Immunol. 6, 1182–1190 (2005).
Saas, P. et al. TWEAK stimulation of astrocytes and the proinflammatory consequences. Glia 32, 102–107 (2000).
Chicheportiche, Y. et al. Proinflammatory activity of TWEAK on human dermal fibroblasts and synoviocytes: blocking and enhancing effects of anti-TWEAK monoclonal antibodies. Arthritis Res. 4, 126–133 (2002).
Hosokawa, Y., Hosokawa, I., Ozaki, K., Nakae, H. & Matsuo, T. Proinflammatory effects of tumour necrosis factor-like weak inducer of apoptosis (TWEAK) on human gingival fibroblasts. Clin. Exp. Immunol. 146, 540–549 (2006).
Kamata, K. et al. Involvement of TNF-like weak inducer of apoptosis in the pathogenesis of collagen-induced arthritis. J. Immunol. 177, 6433–6439 (2006).
Mueller, A. M. et al. Targeting fibroblast growth factor-inducible-14 signaling protects from chronic relapsing experimental autoimmune encephalomyelitis. J. Neuroimmunol. 159, 55–65 (2005).
Desplat-Jego, S. et al. Anti-TWEAK monoclonal antibodies reduce immune cell infiltration in the central nervous system and severity of experimental autoimmune encephalomyelitis. Clin. Immunol. 117, 15–23 (2005).
Winkles, J. A., Tran, N. L. & Berens, M. E. TWEAK and Fn14: new molecular targets for cancer therapy? Cancer Lett. 235, 11–17 (2005).
Watts, G. S. et al. Identification of Fn14/TWEAK receptor as a potential therapeutic target in esophageal adenocarcinoma. Int. J. Cancer 121, 2132–2139 (2007).
Xu, L. G. & Shu, H. B. TNFR-associated factor-3 is associated with BAFF-R and negatively regulates BAFF-R-mediated NF-κB activation and IL-10 production. J. Immunol. 169, 6883–6889 (2002).
Fotin-Mleczek, M. et al. Tumor necrosis factor receptor-associated factor (TRAF) 1 regulates CD40-induced TRAF2-mediated NF-κB activation. J. Biol. Chem. 279, 677–685 (2004).
Hauer, J. et al. TNF receptor (TNFR)-associated factor (TRAF) 3 serves as an inhibitor of TRAF2/5-mediated activation of the noncanonical NF-κB pathway by TRAF-binding TNFRs. Proc. Natl Acad. Sci. USA 102, 2874–2879 (2005).
Trendelenburg, G. et al. Serial analysis of gene expression identifies metallothionein-II as major neuroprotective gene in mouse focal cerebral ischemia. J. Neurosci. 22, 5879–5888 (2002).
Eming, S. A., Krieg, T. & Davidson, J. M. Inflammation in wound repair: molecular and cellular mechanisms. J. Invest. Dermatol. 127, 514–525 (2007).
Werner, S. & Grose, R. Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 83, 835–870 (2003).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
de Visser, K. E., Eichten, A. & Coussens, L. M. Paradoxical roles of the immune system during cancer development. Nature Rev. Cancer 6, 24–37 (2006).
Winkles, J. A. et al. Role of TWEAK and Fn14 in tumor biology. Front. Biosci. 12, 2761–2771 (2007).
Kaduka, Y. et al. TWEAK mediates anti-tumor effect of tumor-infiltrating macrophage. Biochem. Biophys. Res. Commun. 331, 384–390 (2005).
Witz, I. P. Yin-yang activities and vicious cycles in the tumor microenvironment. Cancer Res. 68, 9–13 (2008).
Bergers, G. & Benjamin, L. E. Tumorigenesis and the angiogenic switch. Nature Rev. Cancer 3, 401–410 (2003).
Tonini, T., Rossi, F. & Claudio, P. P. Molecular basis of angiogenesis and cancer. Oncogene 22, 6549–6556 (2003).
Lu, H., Ouyang, W. & Huang, C. Inflammation, a key event in cancer development. Mol. Cancer Res. 4, 221–233 (2006).
Albini, A., Tosetti, F., Benelli, R. & Noonan, D. M. Tumor inflammatory angiogenesis and its chemoprevention. Cancer Res. 65, 10637–10641 (2005).
Jakubowski, A. et al. Dual role for TWEAK in angiogenic regulation. J. Cell Sci. 115, 267–274 (2002).
Eccles, S. A. & Welch, D. R. Metastasis: recent discoveries and novel treatment strategies. Lancet 369, 1742–1757 (2007).
Gupta, G. P. & Massague, J. Cancer metastasis: building a framework. Cell 127, 679–695 (2006).
Steeg, P. S. Tumor metastasis: mechanistic insights and clinical challenges. Nature Med. 12, 895–904 (2006).
Hoelzinger, D. B., Demuth, T. & Berens, M. E. Autocrine factors that sustain glioma invasion and paracrine biology in the brain microenvironment. J. Natl Cancer Inst. 99, 1583–1593 (2007).
Paleolog, E. M. Angiogenesis in rheumatoid arthritis. Arthritis Res. 4 (Suppl. 3), S81–S90 (2002).
McInnes, I. B. & Schett, G. Cytokines in the pathogenesis of rheumatoid arthritis. Nature Rev. Immunol. 7, 429–442 (2007).
Rovin, B. H. The chemokine network in systemic lupus erythematous nephritis. Front. Biosci. 13, 904–922 (2008).
Campbell, S., Michaelson, J., Burkly, L. & Putterman, C. The role of TWEAK/Fn14 in the pathogenesis of inflammation and systemic autoimmunity. Front. Biosci. 9, 2273–2284 (2004).
Schwartz, N. et al. Urinary TWEAK and the activity of lupus nephritis. J. Autoimmun. 27, 242–250 (2006).
Hafler, D. A. et al. Multiple sclerosis. Immunol. Rev. 204, 208–231 (2005).
Baxter, A. G. The origin and application of experimental autoimmune encephalomyelitis. Nature Rev. Immunol. 7, 904–912 (2007).
Dirnagl, U., Iadecola, C. & Moskowitz, M. A. Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci. 22, 391–397 (1999).
Huang, J., Upadhyay, U. M. & Tamargo, R. J. Inflammation in stroke and focal cerebral ischemia. Surg. Neurol. 66, 232–245 (2006).
Simard, J. M., Kent, T. A., Chen, M., Tarasov, K. V. & Gerzanich, V. Brain oedema in focal ischaemia: molecular pathophysiology and theoretical implications. Lancet Neurol. 6, 258–268 (2007).
Yepes, M. & Winkles, J. A. Inhibition of TWEAK activity as a new treatment for inflammatory and degenerative diseases. Drug News Perspect. 19, 589–595 (2006).
Yepes, M. TWEAK and the central nervous system. Mol. Neurobiol. 35, 255–265 (2007).
Yepes, M. Tweak and Fn14 in central nervous system health and disease. Front. Biosci. 12, 2772–2781 (2007).
Nash, P.T & Florin, T. H. J. Tumour necrosis factor inhibitors. Med. J. Austr. 183, 205–208 (2005).
Pastan, I., Hassan, R., Fitzgerald, D. J. & Kreitman, R. J. Immunotoxin therapy of cancer. Nature Rev. Cancer 6, 559–565 (2006).
Sun, M. & Fink, P. J. A new class of reverse signaling costimulators belongs to the TNF family. J. Immunol. 179, 4307–4312 (2007).
Hansson, G. K., Robertson, A. K. & Soderberg-Naucler, C. Inflammation and atherosclerosis. Annu. Rev. Pathol. 1, 297–329 (2006).
Munoz-Garcia, B. et al. Fn14 is upregulated in cytokine-stimulated vascular smooth muscle cells and is expressed in human carotid atherosclerotic plaques: modulation by atorvastatin. Stroke 37, 2044–2053 (2006).
Sack, M. Tumor necrosis factor-α in cardiovascular biology and the potential role for anti-tumor necrosis factor-α therapy in heart disease. Pharmacol. Ther. 94, 123–135 (2002).
Gius, D. et al. Profiling microdissected epithelium and stroma to model genomic signatures for cervical carcinogenesis accommodating for covariates. Cancer Res. 67, 7113–7123 (2007).
Almstrup, K. et al. Embryonic stem cell-like features of testicular carcinoma in situ revealed by genome-wide gene expression profiling. Cancer Res. 64, 4736–4743 (2004).
Hamill, C. A., Michaelson, J. S., Hahm, K., Burkly, L. C. & Kessler, J. A. Age-dependent effects of TWEAK/Fn14 receptor activation on neural progenitor cells. J. Neurosci. Res. 85, 3535–3544 (2007).
Felli, N. et al. Multiple members of the TNF superfamily contribute to IFN-γ-mediated inhibition of erythropoiesis. J. Immunol. 175, 1464–1472 (2005).
Kaplan, M. J. et al. The apoptotic ligands TRAIL, TWEAK, and Fas ligand mediate monocyte death induced by autologous Lupus T cells. J. Immunol. 169, 6020–6029 (2002).
Ren, R. et al. Gene expression profiles identify a role for cyclooxygenase 2-dependent prostanoid generation in BMP6-induced angiogenic responses. Blood 109, 2847–2853 (2007).
Acknowledgements
I thank my laboratory manager S. Brown and all of my other laboratory colleagues for their important contributions to our TWEAK–Fn14 studies. I also thank my external TWEAK–Fn14 research collaborators (in particular, M. Berens of the Translational Genomics Research Institute and M. Yepes of Emory University School of Medicine) for involving my laboratory in their various projects. I am also grateful to A. Keegan, M. Williams and three anonymous referees for their helpful comments on this article. Research in the author's laboratory is supported by NIH grants R01-HL39727 and R01-NS55126 and Susan G. Komen for the Cure grant BCTR0503968.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author has applied for patents describing methods for therapeutic inhibition of Fn14 signalling in diseased tissues.
Related links
Related links
DATABASES
OMIM
FURTHER INFORMATION
Glossary
- Type II transmembrane protein
-
An integral membrane protein that is composed of an amino-terminal intracellular region, a transmembrane domain and a carboxy-terminal extracellular region (for example, Ly49).
- Type I transmembrane protein
-
An integral membrane protein that is composed of an amino-terminal extracellular region, a transmembrane domain and a carboxy-terminal intracellular region (for example, T-cell receptors).
- Type III transmembrane protein
-
An integral membrane protein with a similar orientation as a Type I protein but lacking a signal peptide (for example, B-cell maturation antigen (BCMA)).
Rights and permissions
About this article
Cite this article
Winkles, J. The TWEAK–Fn14 cytokine–receptor axis: discovery, biology and therapeutic targeting. Nat Rev Drug Discov 7, 411–425 (2008). https://doi.org/10.1038/nrd2488
Issue Date:
DOI: https://doi.org/10.1038/nrd2488
This article is cited by
-
Development and validation of a glioma-associated mesenchymal stem cell-related gene prognostic index for predicting prognosis and guiding individualized therapy in glioma
Stem Cell Research & Therapy (2023)
-
Novel Drug Targets for Atrial Fibrillation Identified Through Mendelian Randomization Analysis of the Blood Proteome
Cardiovascular Drugs and Therapy (2023)
-
Advancing Treatment in Bullous Pemphigoid: A Comprehensive Review of Novel Therapeutic Targets and Approaches
Clinical Reviews in Allergy & Immunology (2023)
-
Fn14 exacerbates acute lung injury by activating the NLRP3 inflammasome in mice
Molecular Medicine (2022)
-
Impaired bidirectional communication between interneurons and oligodendrocyte precursor cells affects social cognitive behavior
Nature Communications (2022)