Abstract
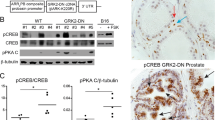
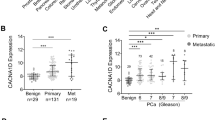
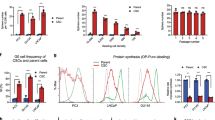
Although prostate cancer cells are often initially sensitive to androgen ablation, they eventually lose this response and continue to survive, grow and spread in the absence of androgenic steroids. The mechanism(s) that underlie resistance to androgen ablation therapy remain mostly unknown. We have demonstrated that elevated caveolin protein levels are associated with human prostate cancer progression in pathological specimens 1 . Here we show that suppression of caveolin expression by a stably transfected antisense caveolin-1 cDNA vector converted androgen-insensitive metastatic mouse prostate cancer cells to an androgen-sensitive phenotype. Orthotopically grown tumors and low-density cell cultures derived from antisense caveolin clones had increased apoptosis in the absence of androgenic steroids, whereas similarly grown tumors and cells from vector (control) clones and parental cells were not sensitive to androgens. Studies using a representative antisense caveolin clone showed that selection for androgen resistance in vivo correlated with increased caveolin levels, and that adenovirus-mediated caveolin expression blocked androgen sensitivity. Our results identify a new candidate gene for hormone-resistant prostate cancer in man and indicate that androgen insensitivity can be an inherent property of metastatic prostate cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Yang, G. et al. Elevated expression of caveolin is associated with prostate and breast cancer. Clin. Cancer Res. 8, 1873 –1880 (1998).
Scardino, P.T. Early detection of prostate cancer. Urol. Clin. North Am. 16, 635–655 (1989).
Huggins, C. & Hodges, C.V. Studies on prostate cancer: effect of castration, of estrogen, and of androgen injection on serum phosphatase in metastatic carcinoma of the prostate. Cancer Res. 1, 293–397 (1941).
McDonnell, T.J. et al. Expression of the protooncogene bcl-2 in the prostate and its association with emergence of androgen-independent prostate cancer. Cancer Res. 52, 6940–6944 (1992).
Colombel, M. et al. Detection of the apoptosis-suppressing oncoprotein bcl-2 in hormone-refractory human prostate cancers. Am. J. Pathol. 143, 390–400 (1993).
Krajewaka, M. et al. Immunohistochemical analysis of bcl-2, bax, bcl-X and mcl-1 expression in prosate cancers. Am. J. Pathol. 148, 1567–1576 (1996).
Furuya, Y., Krajewski, S., Epstein, J.I., Reed, T.C. & Isaacs, J. Expression of bcl-2 and the progression of human and rodent prostatic cancers. Clin. Cancer Res. 2, 389–398 (1996).
Harder T. & Simons K. Caveolae, DIGs, and the dynamics of sphingolipid-cholesterol microdomains. Curr. Opin. Cell. Biol. 9, 534–542 ( 1997).
Fielding, J., Bist, A. & Fielding, P. E. Caveolin mRNA levels are up-regulated by free cholesterol and down-regulated by oxysterols in fibroblast monolayers. Proc. Natl. Acad. Sci. USA 94, 3753–3758 (1997).
Giovannucci, E. et al. A prospective study of dietary fat and risk of prostate cancer. J. Natl. Cancer Inst. 85, 1571– 1579 (1993).
Thompson, T.C. et al. Loss of p53 function leads to metastasis in ras+myc -initiated mouse prostate cancer. Oncogene 10, 869–879 (1995).
Hall, S.J. & Thompson, T.C. Spontaneous metastatic activities but not experimental metastatic activities differentiate primary tumor-derived versus metastasis-derived mouse prostate cancer cell lines. Clin. Exp. Metastasis 15, 484–493 (1997).
Yang, G., Wheeler, T.M., Kattan, M.W., Scardino, P.T. & Thompson, T.C. Perineural invasion of prostate carcinoma cells is associated with reduced apoptotic index. Cancer 78, 1267–1271 ( 1996).
Gavrieli, Y., Sherman, Y. & Ben-Sasson, S.A. Identification of programmed cell death in situ via specific labeling of nuclear DNA fragmentation. J. Cell. Biol. 119, 493–501 ( 1992).
Bett, A.J., Haddara, W., Prevec, L. & Graham, F.L. An efficient and flexible system for construction of adenovirus vectors with insertions or deletions in early regions 1 and 3. Proc. Natl. Acad. Sci. USA 91, 8802–8806 ( 1994).
Acknowledgements
We thank G. Hull, M. McCurdy, S. Shimura and J. Hu for assistance with animal studies; X. Ji for cell culture assistance and M. Marcelli for antiserum and comments on the manuscript. This work was supported by grants from the NIH (CA68814, CA50588 and SPORE P50-58204) and CaPCURE award.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nasu, Y., Timme, T., Yang, G. et al. Suppression of caveolin expression induces androgen sensitivity in metastatic androgen-insensitive mouse prostate cancer cells. Nat Med 4, 1062–1064 (1998). https://doi.org/10.1038/2048
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/2048
This article is cited by
-
PTRF/cavin-1 neutralizes non-caveolar caveolin-1 microdomains in prostate cancer
Oncogene (2014)
-
Caveola-forming proteins caveolin-1 and PTRF in prostate cancer
Nature Reviews Urology (2013)
-
Prostate cancer progression after androgen deprivation therapy: mechanisms of castrate resistance and novel therapeutic approaches
Oncogene (2013)
-
The role of caveolin-1 in prostate cancer: clinical implications
Prostate Cancer and Prostatic Diseases (2010)
-
Growth of hormone-dependent MCF-7 breast cancer cells is promoted by constitutive caveolin-1 whose expression is lost in an EGF-R-mediated manner during development of tamoxifen resistance
Breast Cancer Research and Treatment (2010)