Abstract
Purpose
Ewing’s sarcoma (ES) is a rare and recalcitrant disease which is in need of a development of a novel effective therapy. The aim of this study was to investigate the efficacy of regorafenib on an ES tumor in a patient-derived orthotopic xenograft (PDOX) model.
Methods
The ES PDOX models were established orthotopically in the right chest wall of nude mice to match the site of the tumor in the donor patient. The ES PDOX models were randomized into three groups (G) when the tumor volume reached 75 mm3: G1: untreated control; G2: doxorubicin (DOX) (i.p., 3 mg/kg, weekly, 2 weeks); G3: regorafenib (REG) (p.o., 30 mg/kg, daily, 2 weeks). Tumor volume and body weight were measured twice a week. All mice were sacrificed on day 15.
Results
DOX was ineffective compared to the control group (P = 0.229). REG regressed the tumor size (P < 0.001 and P < 0.001, relative to control and DOX, respectively).
Conclusions
Our findings suggest that REG has clinical potential for ES patients whose tumors respond to REG in a PDOX model.
Similar content being viewed by others
Introduction
Ewing’s sarcoma (ES) is an aggressive, poorly differentiated tumor derived from bone and/or soft tissue. ES is the second most common primary bone tumor, which occurs mainly in children and adolescents [1]. The overall survival (OS) of ES patients with localized disease has improved up to approximately 70% with the development of multidisciplinary treatment [2]. However, the OS of ES patients with metastases and/or recurrence is still very low [3]. Thus, more effective therapy is needed to improve the outcome of ES patients.
Regorafenib (REG) is a multi-tyrosine-kinase inhibitor that binds to the following receptors: platelet-derived growth factor receptor (PDGFRB), vascular endothelial growth factor receptors (VEGFR), angiopoietin-1 receptor, fibroblast growth factor receptor 1 (FGFR1) and mutant kinases such as c-KIT [4]. Initially, REG was approved for patients with advanced colorectal cancer based on previous clinical trials which showed an OS benefit [5,6,7]. Subsequently, REG was approved for patients with gastrointestinal stromal tumors after progression and as standard treatment for hepatocellular carcinoma as a second-line therapy [8,9,10,11]. A randomized double-blind, placebo-controlled, Phase II of REG showed improved progression-free survival (PFS) in non-adipocytic soft-tissue sarcomas [7]. REG improved the quality-adjusted survival for patients with DOX-pretreated soft-tissue sarcoma (STS) [12]. REG showed clinical antitumor efficacy in metastatic osteosarcoma [13], REG was effective in metastatic biliary tract adenocarcinoma [14], glioblastoma [15], lung squamous-cell carcinoma [16], multiple myeloma [17], and adenoid-cystic carcinomas [18].
Previously, we have developed many types of PDOX nude mouse models using the surgical orthotopic implantation (SOI) technique [19]. The PDOX models can be metastatic and mimic the original patient’s tumor, contrary to the classical subcutaneous model. We previously developed the chest-wall ES PDOX model with a rare genetic alteration which is FUS-ERG fusion and cyclin-dependent kinase 2A/B deletion [20]. We identified clinically effective therapy for bone marrow and organ metastases using the ES PDOX model [21, 22]. Bone marrow and organ metastases responded the regimens identified in the PDOX models, and the OS of the patient was prolonged. The PDOX models can also be utilized as an alternative to clinical trials which can save time and money, although further large-scale studies are warranted to confirm the concordance of clinical and PDOX drug response.
The aim of this study was to investigate the efficacy of REG on a chest wall ES using the ES PDOX model.
Materials and methods
Animals
We performed this study using 4–6 weeks old athymic nu/nu female nude mice (AntiCancer, Inc., San Diego, CA). All animals were maintained under the high efficiency particulate arrestance (HEPA)-filtered racks under standard conditions of 12-h light/dark cycles. All experiments were performed with an AntiCancer Institutional Animal Care and Use Committee (IACUC)-protocol specifically approved for this study and in accordance with the principles and procedures outlined in the National Institutes of Health Guide for the Care and Use of Animals under Assurance Number A3873-1. All animal procedures were conducted as we described before [20].
Establishment of the ES PDOX model
This ES tumor originally occurred in the right chest wall of a patient. Tumor specimens were previously brought to AntiCancer, Inc. from the Department of Surgery, University of California, Los Angeles (UCLA) when the patient received curative-intent surgery. The patient signed an informal consent form. This study was approved by the Institutional Review Board of UCLA. Tumor specimens were initially implanted into nude mice subcutaneously. The subcutaneous tumors, grown in nude mice, were harvested and cut into 2 mm-diameter fragments. The fragments were implanted into the right chest wall of nude mice by SOI technique, as described previously [21].
Treatment protocol for the ES PDOX models
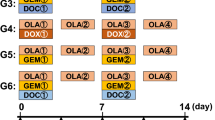
The treatment protocol is illustrated in Fig. 1. The ES PDOX models were randomly divided into 3 groups (G) when the tumor volume reached 80 mm3; G1: untreated control; G2: DOX (i.p., 3 mg/kg, weekly, 2 weeks); G3: REG (REG: p.o., 30 mg/kg, daily, 2 weeks). All regimens were determined by considering previous reports [20, 23]. Tumor size and body weight were measured twice a week. Tumor volume was calculated by the following formula: tumor volume (mm3) = length (mm) × width (mm) × width (mm) × 1/2. All mice were sacrificed on day 15.
Histological findings
Tumors were fixed in 10% formalin and embedded in paraffin. Sections were cut at 5 µm and deparaffinized in xylene and rehydrated in an ethanol series. Hematoxylin and eosin (H&E)-staining was performed by our standard protocol [24]. Histopathology was observed using a BHS system microscope (Olympus, Japan). Pathological response to treatment was evaluated in accordance with a previous report [25].
Statistical analysis
All data were analyzed statistically by free statistical software EZR (Saitama Medical Center, Jichi Medical University) [26]. This free software is a graphical-user interface for R (The R Foundation for Statistical Computing, version 3. 4. 1.). EZR is a modified version of R commander (version 2. 4-0) which includes statistical functions for biostatistics. All statistical analyses were performed as we previously reported [27]. Probability values less than 0.05 were considered as statistical significance.
Results
Quantitative treatment efficacy
The treatment response transition of the tumor volume ratio is shown in Fig. 2a. DOX did not have significant efficacy compared to the control group (P = 0.229), matching the previous results in the patients [21, 22]. REG regressed tumor growth relative to the control and the DOX group (P < 0.001 and P < 0.001, respectively). The final tumor volume ratios (day 15/day 0) were as follows: untreated control (G1) (2.55 ± 0.42); DOX (G2) (2.20 ± 0.34); REG (G3) (0.60 ± 0.12). Representative images of the ES PDOX models from each group on day 15 are shown in Fig. 2b–d.
Treatment response of the tumor volume ratio. Line graphs illustrate the tumor volume ratio (tumor volume at each day/day 0). REG significantly regressed the tumor compared to the control group and the DOX group (P < 0.001 and P < 0.001, respectively, N = 6). *P < 0.001. Error bars: ± 1 SD. B–D. Representative images of the ES PDOX mice from each group on day 15. The area circled by black broken line is the tumor treated by each drug. Scale bar is 10 mm
Effect of treatment on body weight
The body weight ratio is shown in Fig. 3. No significant differences were observed among groups on any day. Significant body weight loss was not detected through the treatment.
Effect of treatment on tumor histology
Figure 4a–c shows representative pathological findings. The tumor in the untreated control group is composed of uniform small round cells and cleared out cytoplasm mimicking the original patient’s tumor, as we previously reported (Fig. 4a) [20]. DOX showed a slight reduction of cellularity (Fig. 4b). REG showed significant reduction of cellularity and extensive necrosis (Fig. 4c). Relative cell density in the tumor was extensively reduced in REG treatment compared to the control and DOX treatment groups (Fig. 5a). The extent of necrosis is quantified in Fig. 5b. There was no significant difference between the control group and the DOX group. REG caused significant broad necrosis compared to control group and the DOX group (P < 0.001 and P < 0.001, respectively). The percent of the necrotic area was as follows: untreated control (G1) (3.33 ± 3.73); DOX (G2) (1.67 ± 2.36); REG (G3) (47.5 ± 5.60).
Effect of treatment on tumor cellularity and necrosis. a Relative cell density in tumor. Extensive reduction of tumor cellularity occurred with REG treatment compared to the control and DOX treatment groups. b There was no significant difference between the control group and the DOX group. REG caused significant broad necrosis compared to the control group and the DOX group (P < 0.001 and P < 0.001, respectively, N = 6). *P < 0.001. Error bars: ± 1 SD
Discussion
We have developed PDOX mouse tumor models for discovery of transformative therapy for recalcitrant cancer including ES [19,20,21,22, 28,29,30]. ES is generally defined by a genetic translocation related to the EWSR1 gene [31]. There are several subtypes with a fusion between EWSR1 gene and the regulatory ETS genes. However, EWSR1-FLI1 fusion accounts for approximately 90% of ES patients, which occurs due to a translocation involving chromosomes 11 and 22 [32]. ES patients with a EWSR1-FLI1 fusion have a heterogeneous response to therapy [33, 34].
The patient donor in the present study had a rare FUS-ERG fusion combined with a CDKN2A deletion, for which there is no standard effective therapy. In our previous studies, we observed that this patient’s PDOX model was sensitive to palbociclib and linsitinib [20]. The patient responded extensively when treatment was based on the PDOX results. Subsequently, we found the patient’s PDOX model was sensitive to irinotecan combined with temozolomide and eribulin [21, 22] and the patient again responded to treatment based on these PDOX results. The patient donor of the ES PDOX model was resistant to DOX, consistent with present and previous PDOX studies of this tumor.
Currently REG is being tested for ES patients in a Phase II clinical trial SARC024 (NCT02048371) which already achieved a primary endpoint of increased PFS [35]. Therefore, REG is expected to be a promising drug for ES patients. However, the efficacy of REG varies in each patient due to heterogeneity of this type of tumor and need to be tested first directly in PDOX models of each patient. The present and previous PDOX studies described above indicate the potential of the PDOX model to identify both ineffective and effective drugs for each patient.
In conclusion, REG regressed the chest wall ES tumor in a PDOX model. Our findings suggest that REG have a potential to be novel effective therapy for ES patients who test positively in a PDOX model.
References
Balamuth NJ, Womer RB (2010) Ewing’s sarcoma. Lancet Oncol 11:184–192
Kovar H, Alonso J, Aman P, Aryee DN, Ban J, Burchill SA, Burdach S, De Alava E, Delattre O, Dirksen U, Fourtouna A, Fulda S, Helman LJ, Herrero-Martin D, Hogendoorn PC, Kontny U, Lawlor ER, Lessnick SL, Llombart-Bosch A, Metzler M, Moriggl R, Niedan S, Potratz J, Redini F, Richter GH, Riedmann LT, Rossig C, Schäfer BW, Schwentner R, Scotlandi K, Sorensen PH, Staege MS, Tirode F, Toretsky J, Ventura S, Eggert A, Ladenstein R (2012) The first European interdisciplinary ewing sarcoma research summit. Front Oncol 2:54
Paulussen M, Ahrens S, Burdach S, Craft A, Dockhorn-Dworniczak B, Dunst J, Fröhlich B, Winkelmann W, Zoubek A, Jürgens H (1998) Primary metastatic (stage IV) Ewing tumor: survival analysis of 171 patients from the EICESS studies. European Intergroup Cooperative Ewing Sarcoma Studies. Annals Oncol 9:275–281
Wilhelm SM, Dumas J, Adnane L, Lynch M, Carter CA, Schütz G, Thierauch KH, Zopf D (2011) Regorafenib (BAY 73-4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer 129:245–255
FDA approves (2012) regorafenib (Stivarga) for metastatic colorectal cancer. Oncology 26:896
Grothey A, Van Cutsem E, Sobrero A, Siena S, Falcone A, Ychou M, Humblet Y, Bouché O, Mineur L, Barone C, Adenis A, Tabernero J, Yoshino T, Lenz HJ, Goldberg RM, Sargent DJ, Cihon F, Cupit L, Wagner A, Laurent D, CORRECT Study Group (2013) Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381:303–312
Li J, Qin S, Xu R, Yau TC, Ma B, Pan H, Xu J, Bai Y, Chi Y, Wang L, Yeh KH, Bi F, Cheng Y, Le AT, Lin JK, Liu T, Ma D, Kappeler C, Kalmus J, Kim TW, CONCUR Investigators (2015) Regorafenib plus best supportive care versus placebo plus best supportive care in Asian patients with previously treated metastatic colorectal cancer (CONCUR): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol 16:619–629
Demetri GD, Reichardt P, Kang YK, Blay JY, Rutkowski P, Gelderblom H, Hohenberger P, Leahy M, von Mehren M, Joensuu H, Badalamenti G, Blackstein M, Le Cesne A, Schöffski P, Maki RG, Bauer S, Nguyen BB, Xu J, Nishida T, Chung J, Kappeler C, Kuss I, Laurent D, Casali PG, GRID study investigators (2013) Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): an international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 381:295–302
Poh A (2017) Regorafenib approved for liver cancer. Cancer Discov 7:660
Duffy AG, Greten TF (2017) Liver cancer: Regorafenib as second-line therapy in hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 14:141–142
Kim K, Jha R, Prins PA, Wang H, Chacha M, Hartley ML, He AR (2017) Regorafenib in advanced hepatocellular carcinoma (HCC): considerations for treatment. Cancer Chemother Pharmacol 80:945–954
Berry V, Basson L, Bogart E, Mir O, Blay JY, Italiano A, Bertucci F, Chevreau C, Clisant-Delaine S, Liegl-Antzager B, Tresch-Bruneel E, Wallet J, Taieb S, Decoupigny E, Le Cesne A, Brodowicz T, Penel N (2017) REGOSARC: regorafenib versus placebo in doxorubicin-refractory soft-tissue sarcoma-A quality-adjusted time without symptoms of progression or toxicity analysis. Cancer 123:2294–2302
Duffaud F, Mir O, Boudou-Rouquette P, Piperno-Neumann S, Penel N, Bompas E, Delcambre C, Kalbacher E, Italiano A, Collard O, Chevreau C, Saada E, Isambert N, Delaye J, Schiffler C, Bouvier C, Vidal V, Chabaud S, Blay JY; French Sarcoma Group (2019) Efficacy and safety of regorafenib in adult patients with metastatic osteosarcoma: a non-comparative, randomised, double-blind, placebo-controlled, phase 2 study. Lancet Oncol 20:120–133
Sun W, Patel A, Normolle D, Patel K, Ohr J, Lee JJ, Bahary N, Chu E, Streeter N, Drummond S (2018) A phase 2 trial of regorafenib as a single agent in patients with chemotherapy-refractory, advanced, and metastatic biliary tract adenocarcinoma. Cancer. https://doi.org/10.1002/cncr.31872
Lombardi G, De Salvo GL, Brandes AA, Eoli M, Rudà R, Faedi M, Lolli I, Pace A, Daniele B, Pasqualetti F, Rizzato S, Bellu L, Pambuku A, Farina M, Magni G, Indraccolo S, Gardiman MP, Soffietti R, Zagonel V (2019) Regorafenib compared with lomustine in patients with relapsed glioblastoma (REGOMA): a multicentre, open-label, randomised, controlled, phase 2 trial. Lancet Oncol 20:110–119
Hu X, Wu LW, Zhang ZY, Chen ML, Li YL, Zhang C (2018) The anti-tumor effect of regorafenib in lung squamous cell carcinoma in vitro. Biochem Biophys Res Commun 503:1123–1129
Breitkreutz I, Podar K, Figueroa-Vazquez V, Wilhelm S, Hayden PJ, Anderson KC, Raab MS (2018) The orally available multikinase inhibitor regorafenib (BAY 73-4506) in multiple myeloma. Ann Hematol 97:839–849
Chen C, Choudhury S, Wangsa D, Lescott CJ, Wilkins DJ, Sripadhan P, Liu X, Wangsa D, Ried T, Moskaluk C, Wick MJ, Glasgow E, Schlegel R, Agarwal S (2017) A multiplex preclinical model for adenoid cystic carcinoma of the salivary gland identifies regorafenib as a potential therapeutic drug. Sci Rep 7:11410
Hoffman RM (2015) Patient-derived orthotopic xenografts: better mimic of metastasis than subcutaneous xenografts. Nat Rev Cancer 15:451–452
Murakami T, Singh AS, Kiyuna T, Dry SM, Li Y, James AW, Igarashi K, Kawaguchi K, DeLong JC, Zhang Y, Hiroshima Y, Russell T, Eckardt MA, Yanagawa J, Federman N, Matsuyama R, Chishima T, Tanaka K, Bouvet M, Endo I, Eilber FC, Hoffman RM (2016) Effective molecular targeting of CDK4/6 and IGF-1R in a rare FUS-ERG fusion CDKN2A-deletion doxorubicin-resistant Ewing’s sarcoma patient-derived orthotopic xenograft (PDOX) nude-mouse model. Oncotarget 7:47556–47564
Miyake K, Murakami T, Kiyuna T, Igarashi K, Kawaguchi K, Li Y, Singh AS, Dry SM, Eckardt MA, Hiroshima Y, Momiyama M, Matsuyama R, Chishima T, Endo I, Eilber FC, Hoffman RM (2018) Eribulin regresses a doxorubicin-resistant Ewing’s sarcoma with a FUS-ERG fusion and CDKN2A-deletion in a patient-derived orthotopic xenograft (PDOX) nude mouse model. J Cell Biochem 119:967–972
Miyake K, Murakami T, Kiyuna T, Igarashi K, Kawaguchi K, Miyake M, Li Y, Nelson SD, Dry SM, Bouvet M, Elliott IA, Russell TA, Singh AS, Eckardt MA, Hiroshima Y, Momiyama M, Matsuyama R, Chishima T, Endo I, Eilber FC, Hoffman RM (2017) The combination of temozolomide-irinotecan regresses a doxorubicin-resistant patient-derived orthotopic xenograft (PDOX) nude-mouse model of recurrent Ewing’s sarcoma with a FUS-ERG fusion and CDKN2A deletion: Direction for third-line patient therapy. Oncotarget 8:103129–103136
Daudigeos-Dubus E, Le Dret L, Lanvers-Kaminsky C, Bawa O, Opolon P, Vievard A, Villa I, Pagès M, Bosq J, Vassal G, Zopf D, Geoerger B (2015) Regorafenib: antitumor activity upon mono and combination therapy in preclinical pediatric malignancy models. PLoS One 10:e0142612
Murakami T, Kiyuna T, Kawaguchi K, Igarashi K, Singh AS, Hiroshima Y, Zhang Y, Zhao M, Miyake K, Nelson SD, Dry SM, Li Y, DeLong JC, Lwin TM, Chishima T, Tanaka K, Bouvet M, Endo I, Eilber FC, Hoffman RM (2017) The irony of highly-effective bacterial therapy of a patient-derived orthotopic xenograft (PDOX) model of Ewing’s sarcoma, which was blocked by Ewing himself 80 years ago. Cell Cycle 16:1046–1052
Wunder JS, Paulian G, Huvos AG, Heller G, Meyers PA, Healey JH (1998) The histological response to chemotherapy as a predictor of the oncological outcome of operative treatment of Ewing sarcoma. J Bone Jt Surg Am 80:1020–1033
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl 48:452–458
Miyake K, Kiyuna T, Miyake M, Zhao M, Wangsiricharoen S, Kawaguchi K, Zhang Z, Higuchi T, Razmjooei S, Li Y, Nelson SD, Russell T, Singh A, Murakami T, Hiroshima Y, Momiyama M, Matsuyama R, Chishima T, Singh SR, Chawla SP, Eilber FC, Endo I, Hoffman RM (2018) Tumor-targeting Salmonella typhimurium A1-R overcomes partial carboplatinum-resistance of a cancer of unknown primary (CUP). Tissue Cell 54:144–149
Kiyuna T, Tome Y, Murakami T, Kawaguchi K, Igarashi K, Miyake K, Miyake M, Li Y, Nelson SD, Dry SM, Singh AS, Russell TA, Elliott I, Singh SR, Kanaya F, Eilber FC, Hoffman RM (2018) Trabectedin arrests a doxorubicin-resistant PDGFRA-activated liposarcoma patient-derived orthotopic xenograft (PDOX) nude mouse model. BMC Cancer 18:840
Kawaguchi K, Miyake K, Han Q, Li S, Tan Y, Igarashi K, Kiyuna T, Miyake M, Higuchi T, Oshiro H, Zhang Z, Razmjooei S, Wangsiricharoen S, Bouvet M, Singh SR, Unno M, Hoffman RM (2018) Oral recombinant methioninase (o-rMETase) is superior to injectable rMETase and overcomes acquired gemcitabine resistance in pancreatic cancer. Cancer Lett 432:251–259
Igarashi K, Kawaguchi K, Li S, Han Q, Tan Y, Murakami T, Kiyuna T, Miyake K, Miyake M, Singh AS, Eckardt MA, Nelson SD, Russell TA, Dry SM, Li Y, Yamamoto N, Hayashi K, Kimura H, Miwa S, Tsuchiya H, Singh SR, Eilber FC, Hoffman RM (2018) Recombinant methioninase in combination with doxorubicin (DOX) overcomes first-line DOX resistance in a patient-derived orthotopic xenograft nude-mouse model of undifferentiated spindle-cell sarcoma. Cancer Lett 417:168–173
Li X, Tanaka K, Nakatani F, Matsunobu T, Sakimura R, Hanada M, Okada T, Nakamura T, Iwamoto Y (2005) Transactivation of cyclin E gene by EWS-Fli1 and antitumor effects of cyclin dependent kinase inhibitor on Ewing’s family tumor cells. Int J Cancer 116:385–394
Delattre O, Zucman J, Plougastel B, Desmaze C, Melot T, Peter M, Kovar H, Joubert I, de Jong P, Rouleau G et al (1992) Gene fusion with an ETS DNA-binding domain caused by chromosome translocation in human tumours. Nature 359:162–165
Parham DM, Hijazi Y, Steinberg SM, Meyer WH, Horowitz M, Tzen CY, Wexler LH, Tsokos M (1999) Neuroectodermal differentiation in Ewing’s sarcoma family of tumors does not predict tumor behavior. Human Pathol 30:911–918
Pinto A, Dickman P, Parham D (2011) Pathobiologic markers of the ewing sarcoma family of tumors: state of the art and prediction of behaviour. Sarcoma 2011:856190
Attia S, Bolejack V, Ganjoo KN et al (2017) A phase II trial of regorafenib (REGO) in patients (pts) with advanced Ewing sarcoma and related tumors (EWS) of soft tissue and bone: SARC024 trial results. J Clin Oncol 35:11005
Acknowledgements
This paper is dedicated to the memory of A. R. Moossa, M.D., and Sun Lee, M.D.
Funding
This work was supported in part by a Yokohama City University research grant “KAMOME Project” which had no role in the research or writing the paper.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. K.M., T.K., K.K., T.H., H.O., Z.Z., S.W., S.R., T.M., Y.H., and RMH are or were unsalaried associates of AntiCancer, Inc. AntiCancer, Inc. uses PDOX models for contract research.
Ethical approval
All experiments were performed with an AntiCancer Institutional Animal Care and Use Committee (IACUC)-protocol specifically approved for this study and in accordance with the principals and procedures outlined in the National Institutes of Health Guide for the Care and Use of Animals under Assurance Number A3873-1.
Informed consent
The patient signed an informed consent form. This study was approved by the Institutional Review Board of UCLA.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miyake, K., Kiyuna, T., Kawaguchi, K. et al. Regorafenib regressed a doxorubicin-resistant Ewing’s sarcoma in a patient-derived orthotopic xenograft (PDOX) nude mouse model. Cancer Chemother Pharmacol 83, 809–815 (2019). https://doi.org/10.1007/s00280-019-03782-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-019-03782-w